|
CH3CHO, C6H5CHO, HCHO (reactivity towards nucleophilic addition reaction). Arrange the following carboxylic acid in their decreasing acidity. Arrange the following compounds in increasing order of their property as indicated. 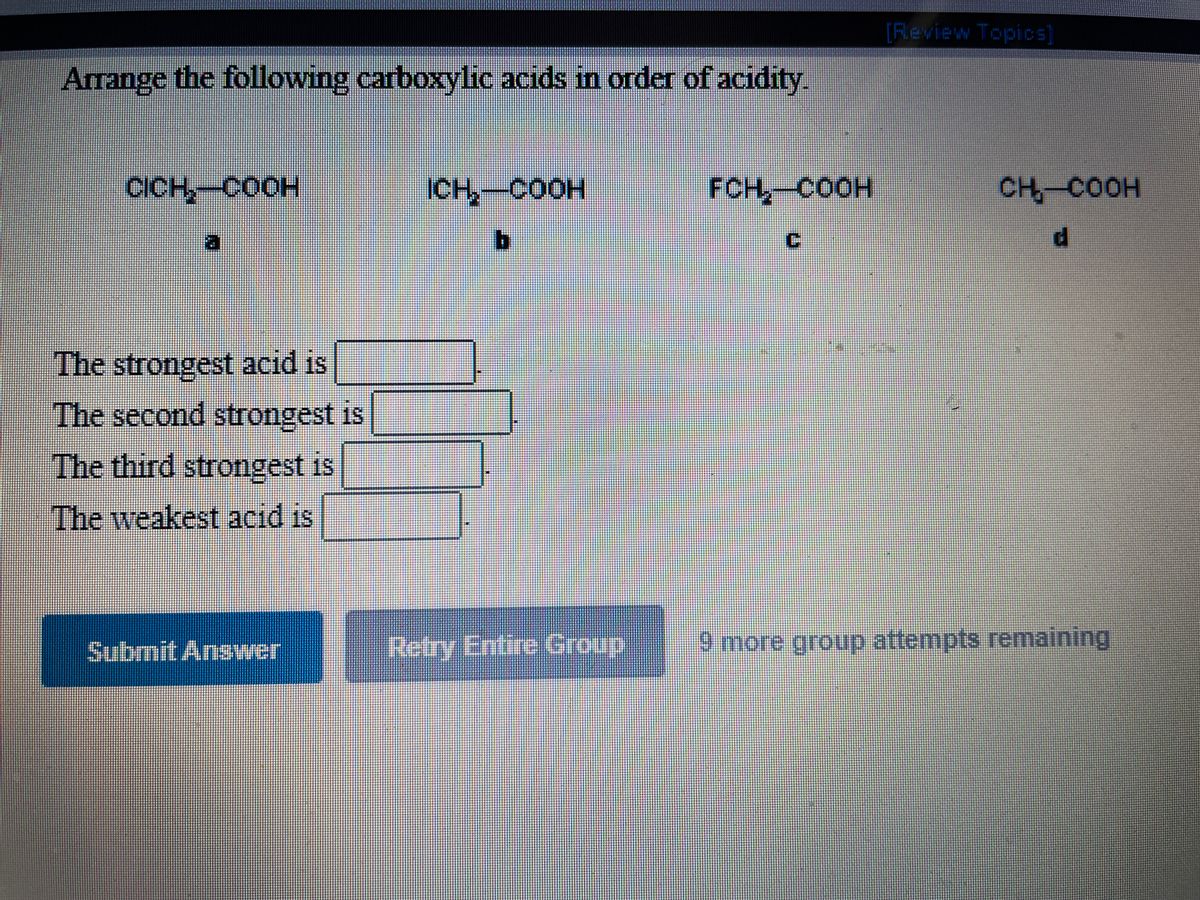 Using the information on this page, you may compare the acidity of carboxylic acid, alcohol, and phenol. Boiling points of carboxylic acids are (a) lower than corresponding alcohols (b). If you have any queries, drop a comment below and we will get back to you. It is possible to compare the acidity of aromatic and aliphatic carboxylic acids. Weve got the study and writing resources you need for your. Start your trial now First week only 4.99 arrowforward. From 1 being the least acidic and 4 being the most acidic, arrange the following organic compounds in terms of increasing acidity. Solution for Ränk the carboxylic acids in order of increasing acidity. Start your trial now First week only 6.99 arrow. We hope this article on ‘Acidity of Carboxylic Acids’ has helped you. Solution for Arrange the following types in order of increasing acidity: carboxylic acids, alcohols, phenols, water. Which is more acidic, alcohol or phenol?Īns: Phenol is more acidic than alcohol since phenoxide ion is more stabilized than alkoxide ion due to the resonance effect. The ranking of these acids in order of increasing acidity is: CH3CH2COOH 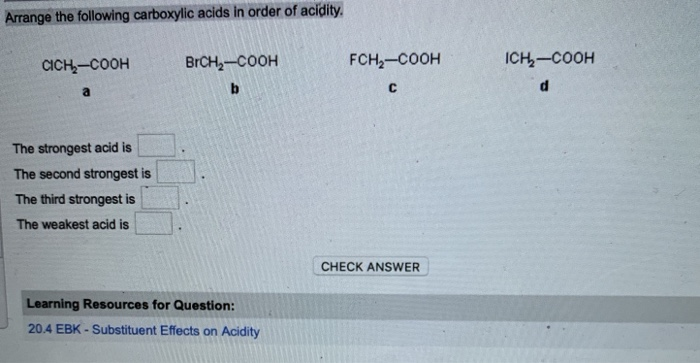 However, in alcohols, no resonance is possible for the alcohols as well as alkoxide ions. The acidity of carboxylic acids is higher in comparison to simple phenols as they react with weak bases like carbonates and bicarbonates to liberate carbon dioxide gas. 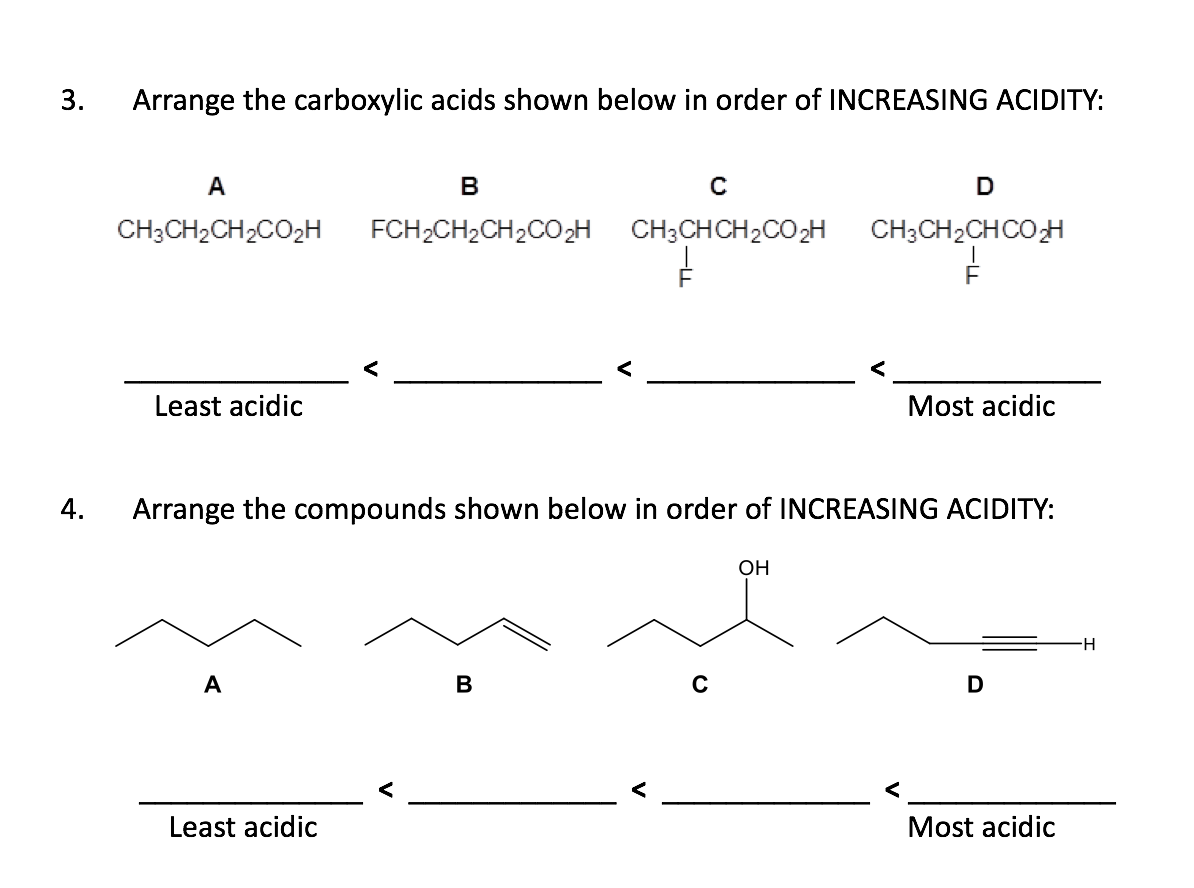 Carbon compounds that contain carboxyl group \(\left( \) forming stable carboxylate ions.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
